Advancing the Future of RNAi Therapeutics
Suzhou Ribo Life Science Co., Ltd. (HKEX: 06938) is a global pioneer in the development of oligonucleotide-based therapeutics, dedicated to advancing RNA interference (RNAi) technology and bringing innovative oligonucleotide drugs to market. Our vertically integrated R&D platform encompasses a comprehensive suite of technologies that support the entire lifecycle of oligonucleotide therapeutics.
With a strong global infrastructure, we have built the capacity to drive oligonucleotide drug innovation worldwide. Our robust siRNA pipeline targets a broad range of diseases—including cardiovascular, metabolic, renal, and liver conditions—with a particular focus on chronic disease treatment. These advancements are driven by our commitment to in-house innovation.


At the Forefront of RNAi Drug Development
Our key technologies are continuously iterating and innovating, supporting the development of our R&D pipeline, with multiple products entering the clinical stage.
-
siRNA Delivery Technologies
Chemical Modification Technologies
Multi-Target Drug Development Platform


At the Forefront of RNAi Drug Development
Our key technologies are continuously iterating and innovating, supporting the development of our R&D pipeline, with multiple products entering the clinical stage.
-

 siRNA Delivery Technologies
siRNA Delivery Technologies
Chemical Modification Technologies
Multi-Target Drug Development Platform -
Therapeutic Areas
Our core therapeutic focus areas address major global health challenges where current treatment options are limited. These areas are characterized by underlying pathogenic mechanisms that align closely with the targeting strengths of our technology platforms.
-
Cardiovascular, Metabolic, and Renal Diseases
-
Liver Diseases
-
Other Therapeutic Areas with High Unmet Medical Need


-


Our Pipeline
We have a robust and diverse pipeline and are at the forefront of oligonucleotide drug innovation. Our development efforts are focused on creating novel therapies for cardiovascular, metabolic, renal, and liver diseases, as well as other areas with significant unmet medical needs.
-


Business Development
We actively pursue global partnerships to accelerate the advancement of our technologies and products, with the goal of delivering the benefits of our RNA-based innovative therapeutics to patients worldwide as swiftly as possible.
We welcome the opportunity to explore collaboration—please feel free to contact us to discuss potential partnerships.
Stay Up to Date with Ribo
-

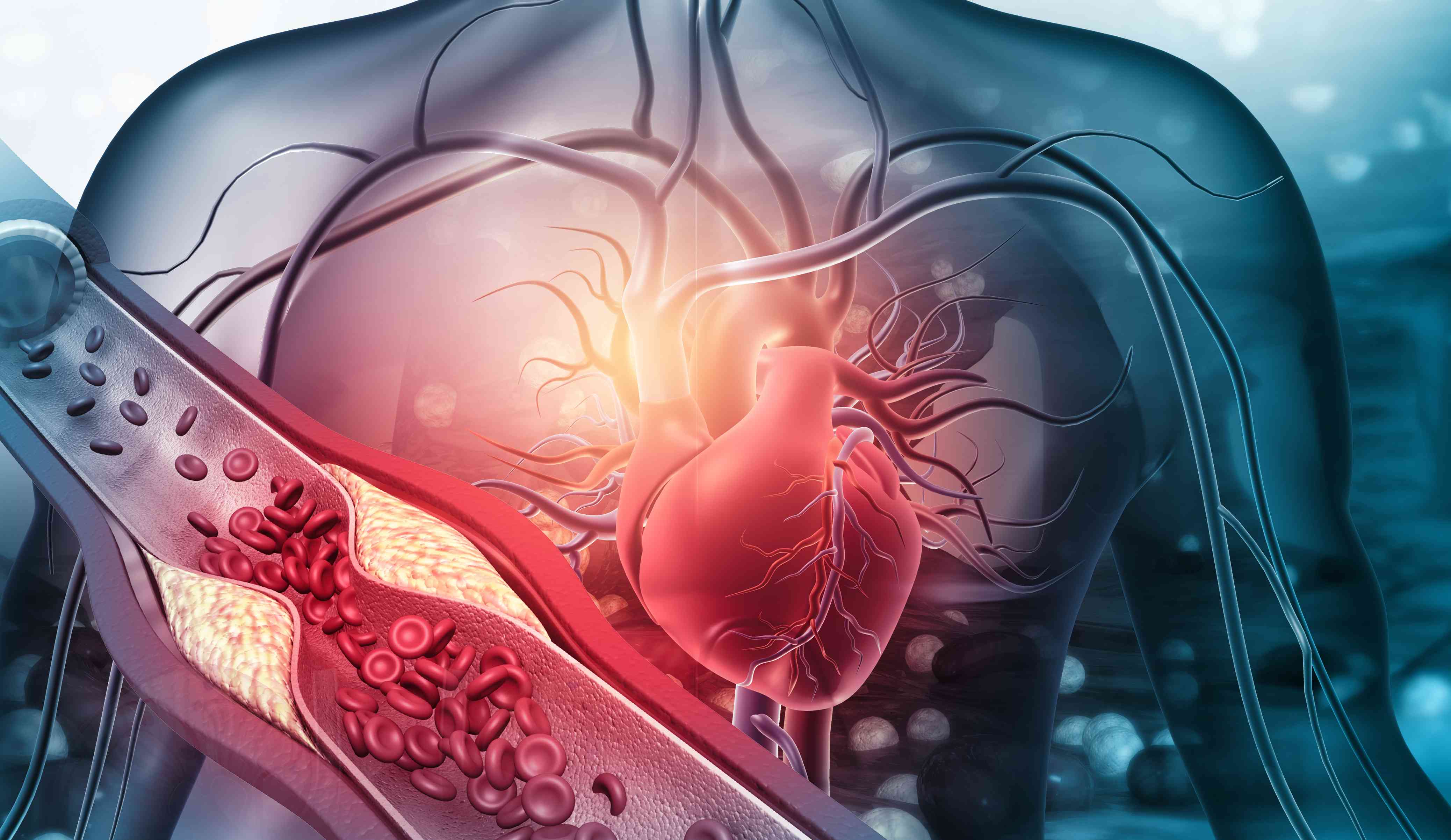
Ribo Submits Phase 2b Clinical Trial Application for world’s first siRNA FXI inhibitor in atrial fibrillation
Ribo Submits Phase 2b Clinical Trial Application for world’s first siRNA FXI inhibitor in atrial fibrillation
Learn more
-


Ribo Appoints CMO and CBO to Empower Global R&D and Partnerships
Ribo Appoints CMO and CBO to Empower Global R&D and Partnerships
Learn more
-


Ribo expands beyond the liver with RiboPepSTAR™ – enabling targeted siRNA delivery to multiple organs
Ribo expands beyond the liver with RiboPepSTAR™ – enabling targeted siRNA delivery to multiple organs
Learn more














